Global Neurology Academy

Reducing the Burden of MS Treatment
Reducing the Burden of MS Treatment: Meeting the Unmet Need
Learning Objectives
- Review the benefit/risk strategies in selecting therapy for MS patients while assessing potential treatment regimens that carry acceptable or diminished risk of disease progression
- Explore emergent concepts in the management of MS, focusing on targeting T- and B-cells including:
- Risks associated with continuous immunosuppression
- Action on the inflammatory activity in the CNS compartment
- Identify strategies that simplify patient dosing and side effects to:
- Increase treatment compliance
- Improve patients’ quality of life
- Slow disease progression
MS is the leading cause for acquired non-traumatic neurological disability in young adults.
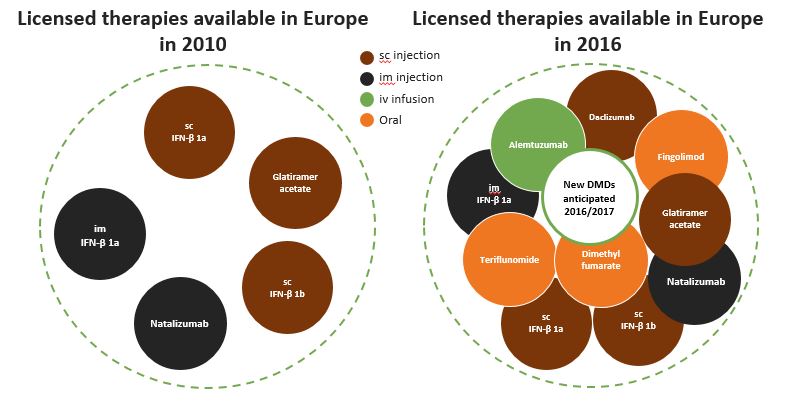
Therapeutic Options in Europe have Doubled in the Last 6 Years
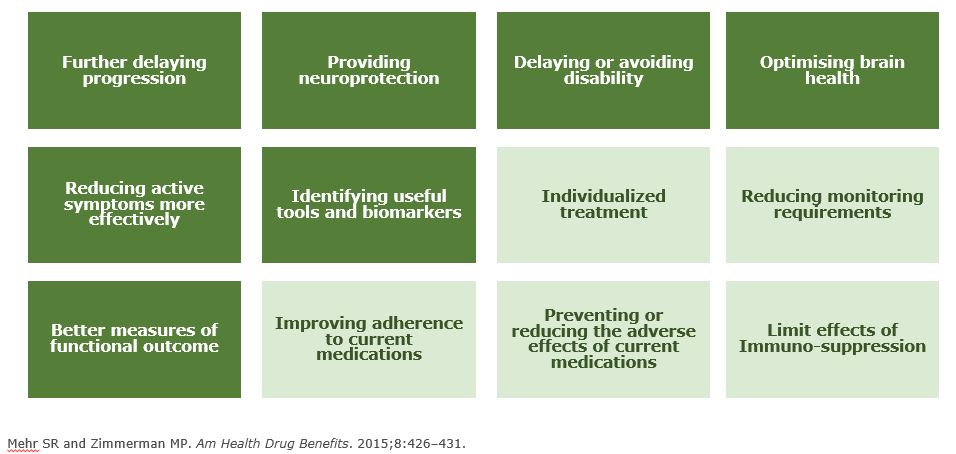
Unmet Needs in MS
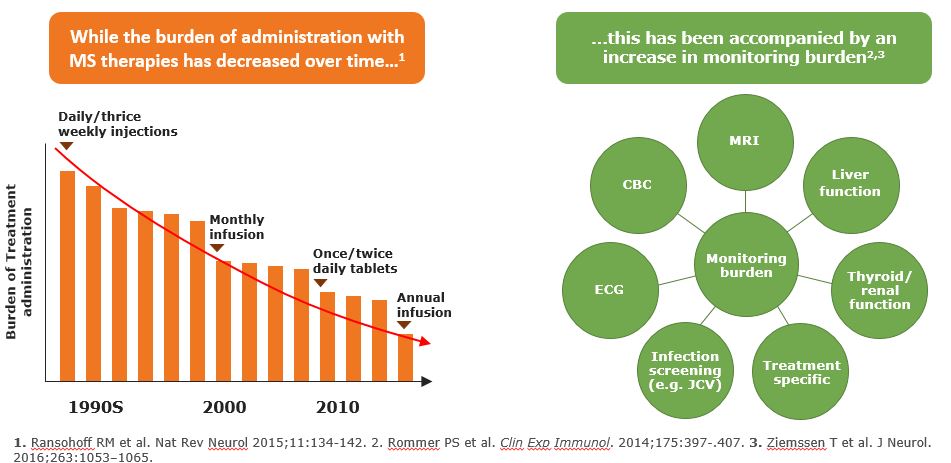
Treatment Burden has Improved – Monitoring Burden Has Increased
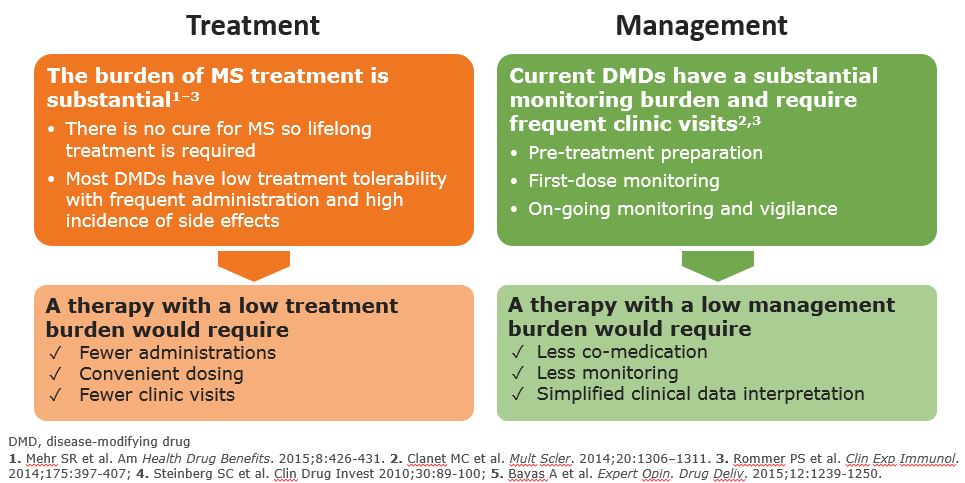
MS Treatment and Management Burden
Platform Therapies Have A Relatively Low Burden Of Monitoring
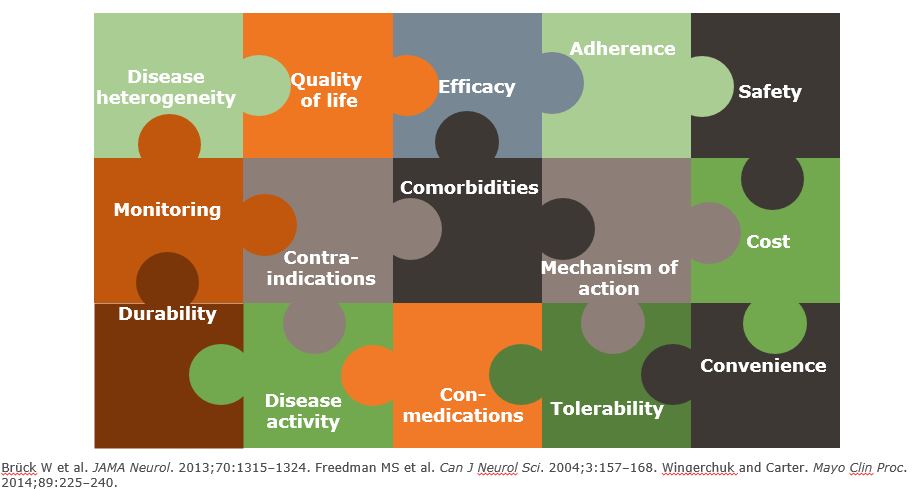
Several factors impact MS treatment decisions
New Pretreatment Paradigm – Treat-2-Target
Treating to achieve NEDA is an emerging treatment paradigm in the management of patients with RMS
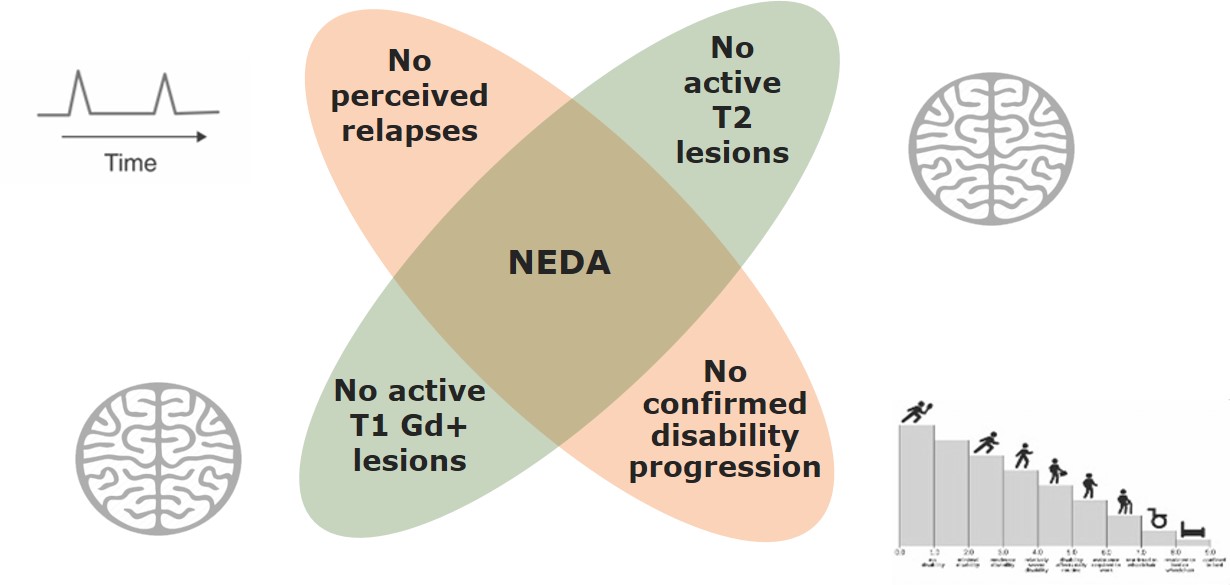
NEDA is based on the principle that relapse rates, disability progression and MRI activity are not independent1–3
Gd+, gadolinium-enhancing; MRI, magnetic resonance imaging; NEDA, no evidence of disease activity; RMS, relapsing MS.
1. Bevan CJ, Cree BA. JAMA. 2014;71:269-270. 2. Sormani MP et al. Mult Scler. 2011;17:541-549. 3. Kappos L et al. Mult Scler J. 2016;22:1297–1305.
Risks Associated with Prolonged or Continuous Immunosuppression
T cells and B cells play critical roles in MS, and therapies targeting lymphocytes have a clinical effect1

1. McFarland HF et al. Nat Immunol. 2007;8:913–919; 2. Nath A, Berger JR. Curr Treat Options Neurol. 2012;14:241–255. 3. Winkelmann A et al. Clin Exp Immunol. 2014;175:425–438.
Malignancy Risk Among MS Treatments
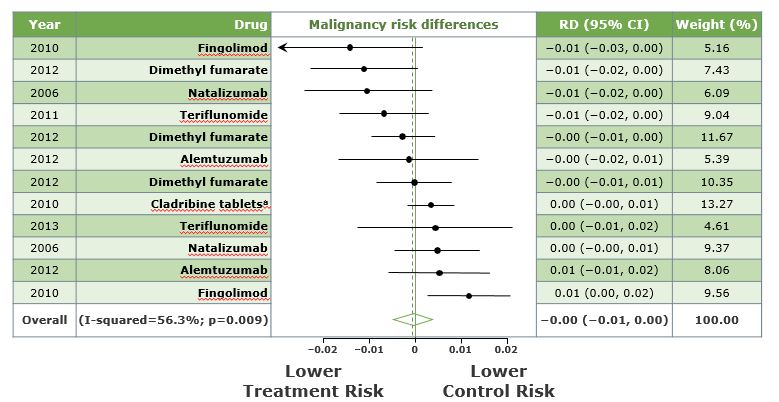
Note: Weights are from random effects analysis.
Risks Associated With Continuous Immunosuppression
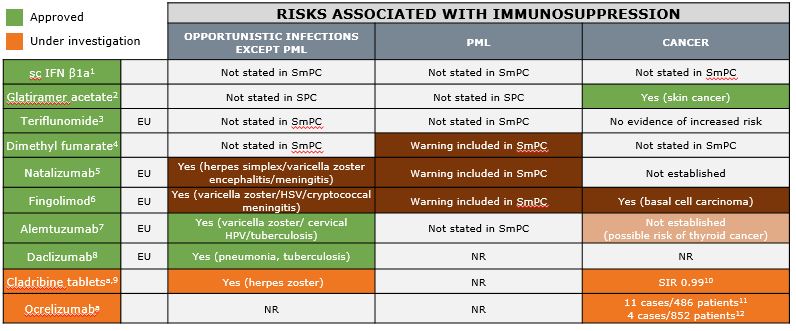
aThese agents are under clinical investigation and have not been proven to be safe and effective. There is no guarantee they will be approved in the sought-after indication. EU = EU label: Medicine is under additional monitoring. HPV, human papillomavirus; HSV, herpes simplex virus; IFN, interferon; NR, not reported; PML, progressive multifocal leukoencephalopathy; sc, subcutaneous; SIR, standardized incidence ratio; SmPC, Summary of Product Characteristics.
1.Rebif® EU SmPC; 2.Copaxone® UK PI; 3.Aubagio® EU SmPC; 4.Tecfidera® EU SmPC; 5.Tysabri® EU SmPC; 6.Gilenya® EU SmPC; 7.Lemtrada® EU SmPC; 8.Zinbryta® EU SmPC; 9.Giovannoni G, et al. N Engl J Med. 2010;362:416–426; 10. Cook S et al. MultScler. 2011;17:578–593; 11. ORATORIO: Montalban X et al. Neurology. 2016;86(Suppl 16):S49.001; 12. OPERA I and II: Hauser SL et al. Neurology. 2016;86(Suppl 16). EU and US labels accessed August 2016.
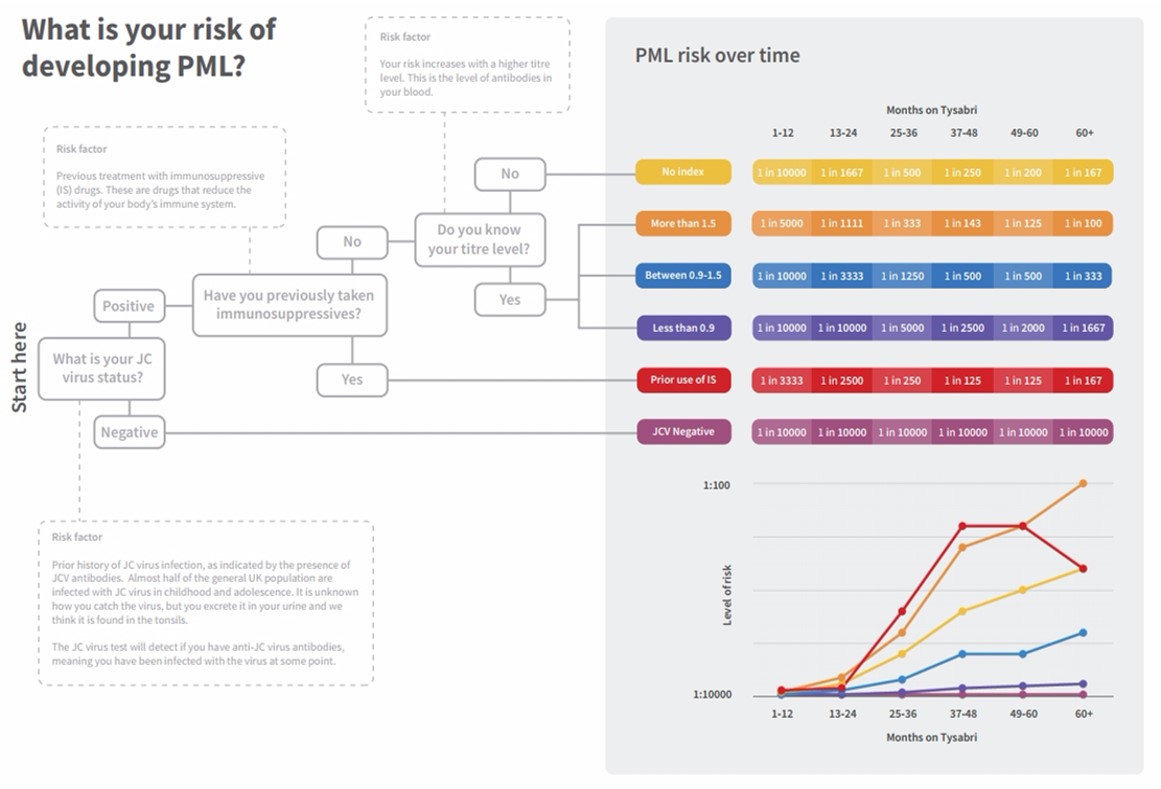
PML Risk Varies over Time with Natalizumab
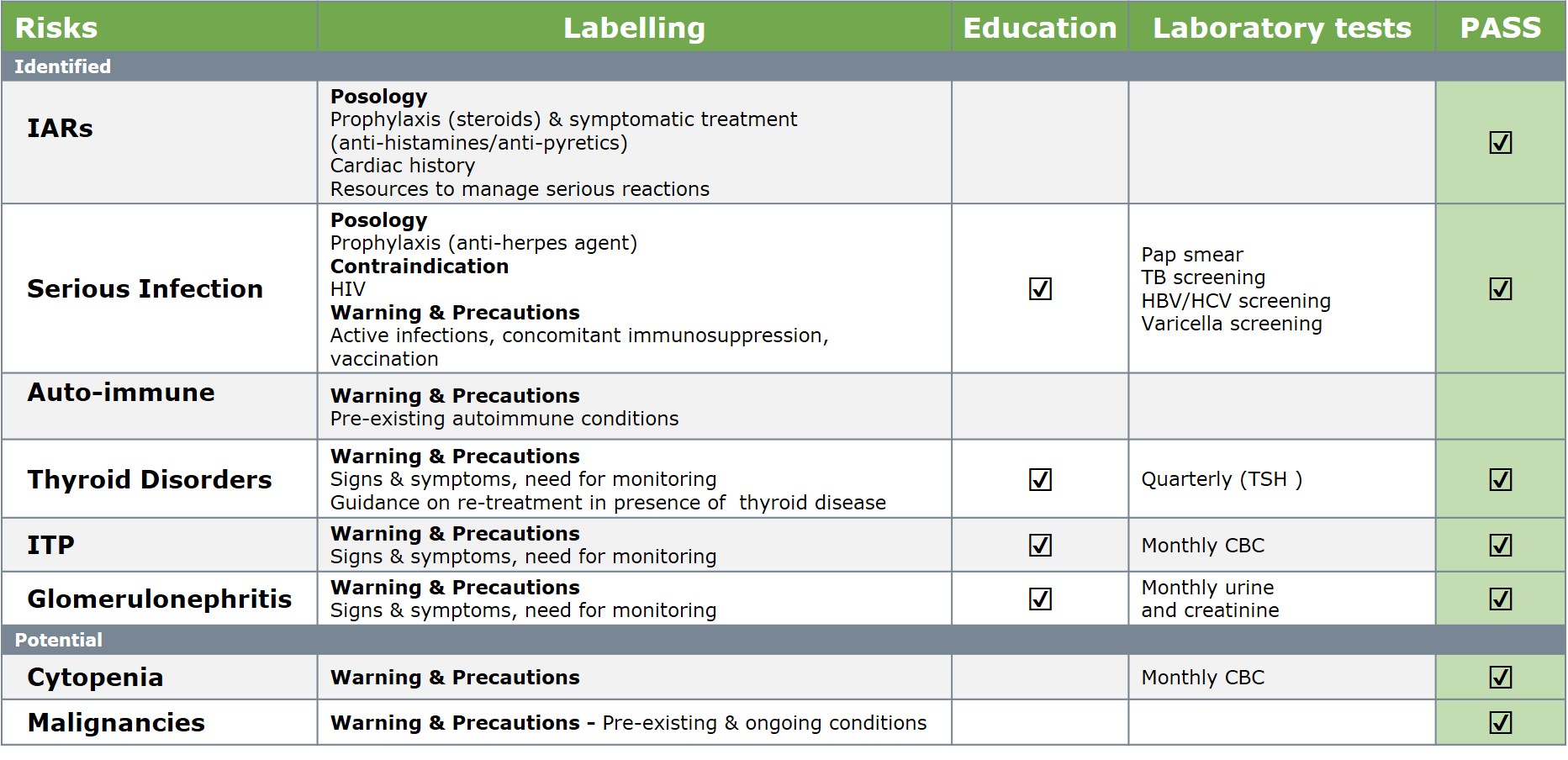
Alemtuzumab Risk Management Strategy
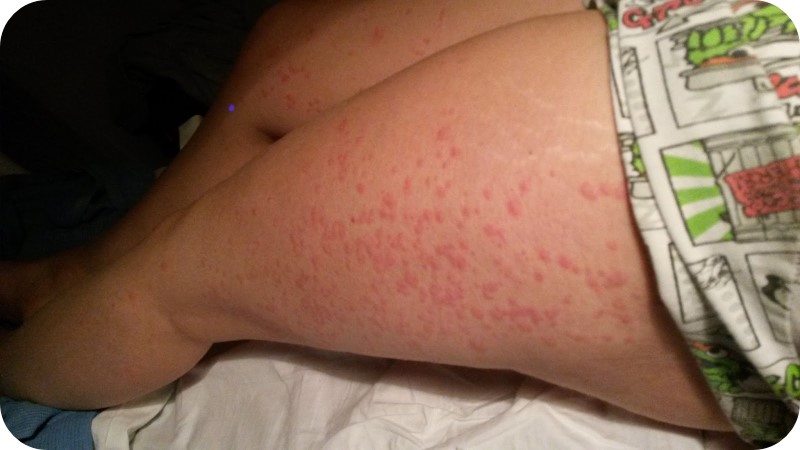
Skin Rash and Angioedema
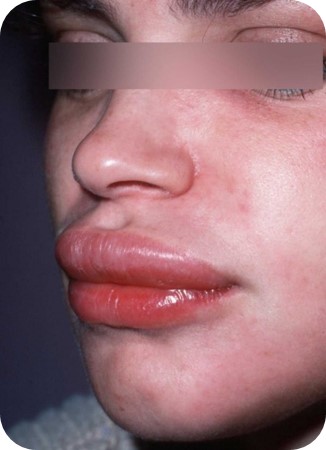
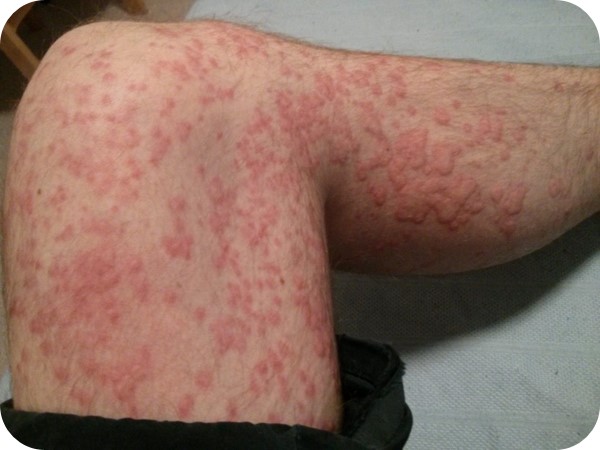
Modified Protocol
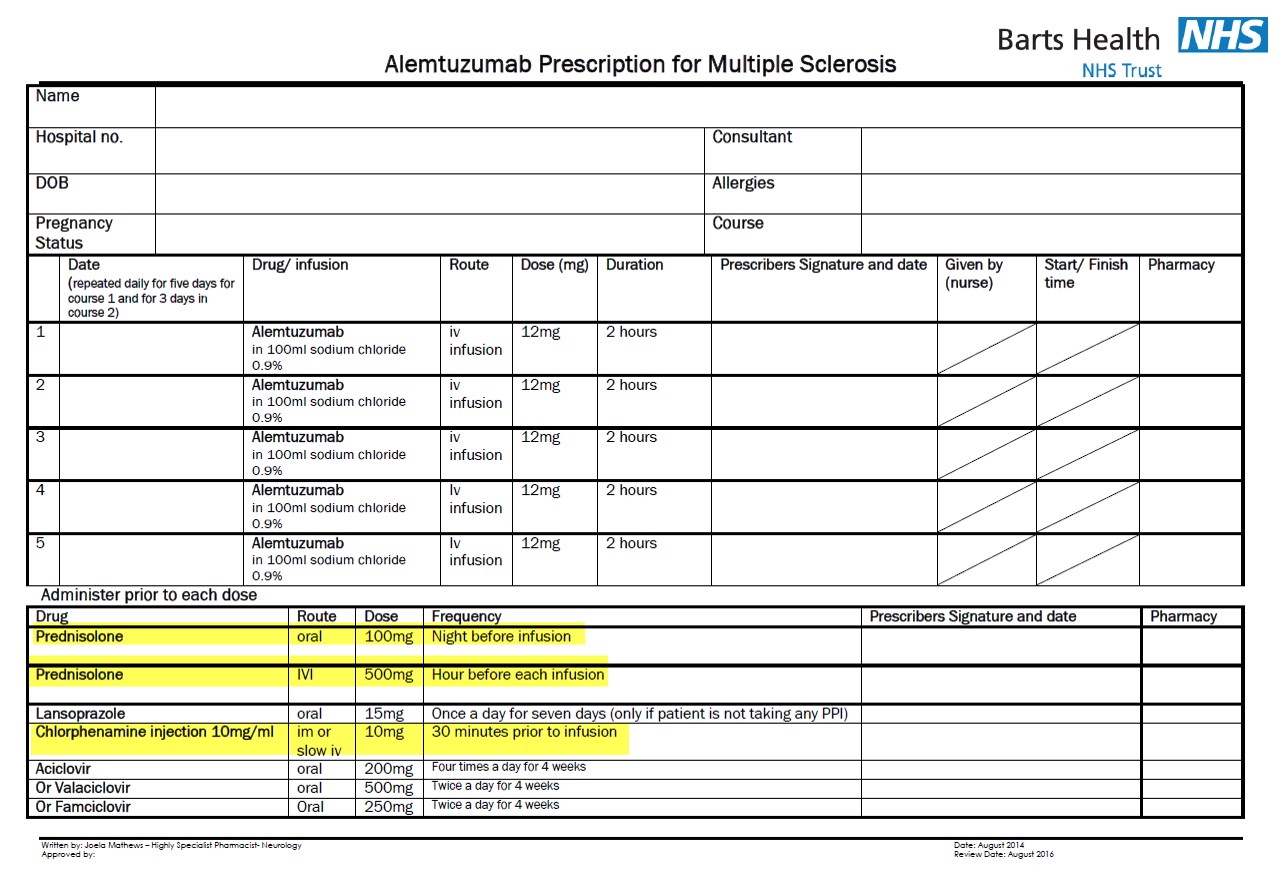
Barts Health NHS. Data on file.
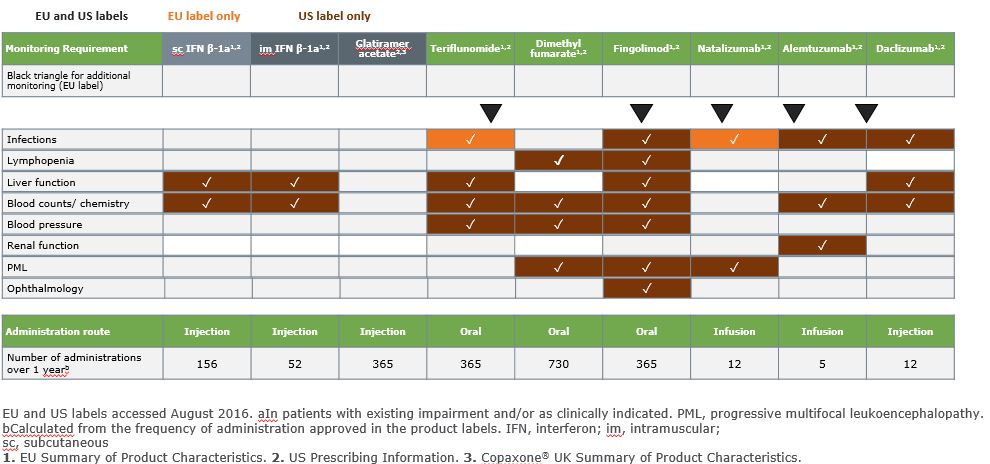
Monitoring Varies by Drug and Duration of Therapy
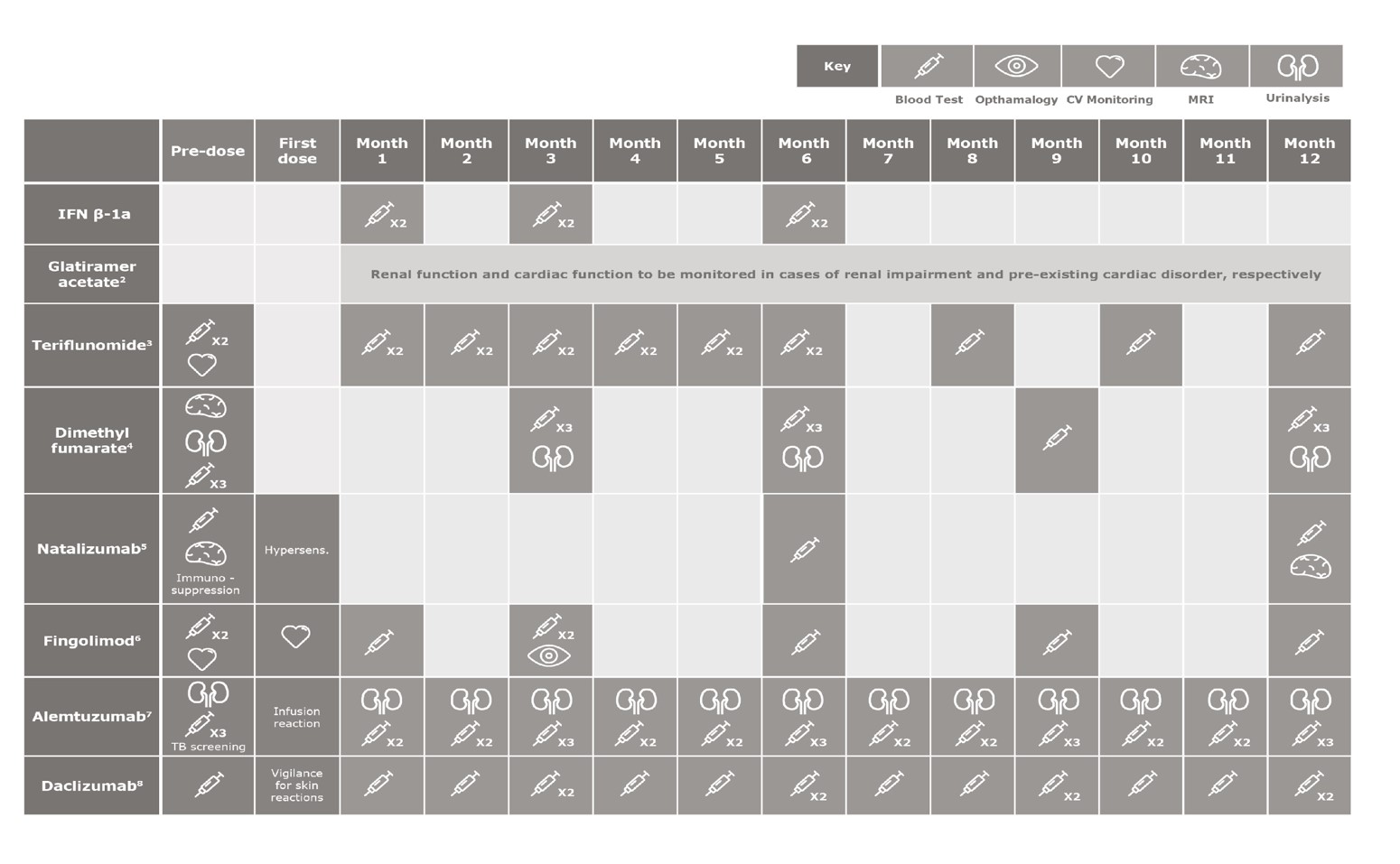
Numbers indicate the number of blood tests. ECG, electrocardiogram; hypersens., hypersensitivity; SmPC, Summary of Product Characteristics.
1.Rebif® EU SmPC; 2.Copaxone® UK PI; 3.Aubagio® EU SmPC; 5.Tysabri® EU SmPC; 6.Gilenya® EU SmPC; 7.Lemtrada® EU SmPC; 8.Zinbryta® EU SmPC.
Patient Adherence
15%–51% of patients with MS do not adhere to their treatment regimen11
61%–96% of patients prefer an intermittent rather than once-daily treatment regimen12
Dose frequency is a major reason why patients with MS miss a dose13
Treatment Frequency Varies by Drug—May Impact Adherence
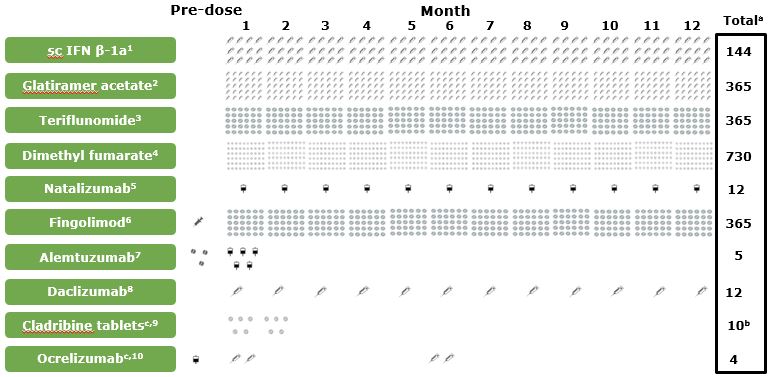
1.Rebif® EU SmPC; 2.Copaxone® SPC; 3.Aubagio® EU SmPC; 4.Tecfidera® EU SmPC; 5.Tysabri® EU SmPC; 6.Gilenya® EU SmPC; 7.Lemtrada® EU SmPC; 8.Zinbryta® EU SmPC; 9.Giovannoni G, et al. N Engl J Med. 2010;362:416–426; 10.Kappos L et al. Lancet. 2011;378:1779–1787; 11.Katsarava Z et al. BMC Neurol. 2015;15:170; 12. Kruk ME, Schwalbe N. ClinTher. 2006;28:1989–1995; 13. Devonshire V et al. Eur J Neurol. 2011;18:69–77.
aTotal number of administrations over the first 12 months of treatment. b3.5 mg/kg. 5 days of treatment separated by 1 month; total number of tablets dependent on weight. cThese agents are under clinical investigation and have not been proven to be safe and effective. There is no guarantee they will be approved in the sought-after indication. IFN, interferon; sc, subcutaneous; SmPC, Summary of Product Characteristics.
Treatment Considerations for MS Must be Personalized to the Patient
- Many factors impact treatment decisions in MS...
- Personal factors
- Prognostic factors
- Disease duration and level of disability
- Risk aversion
- Burden of treatment
- Monitoring requirement
- De-risking strategies, e.g. JCV-testing
- Ideally a therapy should...
- Be effective early in the disease course to maximize long-term outcomes
- Offer durability
- Have a well-characterized long-term safety profile
- Have a low treatment and management burden
This resource is supported by an educational grant from Merck KGaA, Darmstadt, Germany.
Recommended
More Than “Women’s Issues": Women’s Reproductive and Gynaecological Health and Work
More Than “Women’s Issues": Women’s Reproductive and Gynaecological Health and Work
ResourcesMore Than “Women’s Issues": Women’s Reproductive and Gynaecological Health and Work
Now Approved in Europe: First Oral Short-Course Treatment for Highly Active Relapsing MS
Now Approved in Europe: First Oral Short-Course Treatment for Highly Active Relapsing MS
ResourcesNow Approved in Europe: First Oral Short-Course Treatment for Highly Active Relapsing MS
Cladribine Receives Positive CHMP Opinion for Relapsing MS Treatment
Cladribine Receives Positive CHMP Opinion for Relapsing MS Treatment
ResourcesCladribine Receives Positive CHMP Opinion for Relapsing MS Treatment
The Diagnostic Challenges of Spinal Cord Lesions in MS
The Diagnostic Challenges of Spinal Cord Lesions in MS
ResourcesThe Diagnostic Challenges of Spinal Cord Lesions in MS









